
New Indication Added! Jiangsu Vcare’s Second-Generation Highly Selective JAK1 Inhibitor VC005 Tablets Approved for IND in Oral Treatment of Vitiligo
Published Time:
2025-05-09 17:38
Source:
On May 8, 2025, Jiangsu Vcare Pharmatech Co., Ltd. (Jiangsu Vcare) achieved another major milestone in its self-developed second-generation highly selective JAK1 inhibitor, VC005 tablets. The project, intended for the oral treatment of non-segmental vitiligo received official IND approval from the NMPA (Application No.: CXHL2500200).
The VC005 project demonstrates picomolar-level inhibition of JAK1 in vitro, providing a potent mechanism for reversing vitiligo progression. Whigh selectivity towards JAK1 than JAK2, it is expected to mitigate hematological side effects caused by excessive JAK2 inhibition. Additionally, VC005’s inhibitory effects on JAK3 and TYK2 synergize to enhance efficacy, particularly by significantly suppressing the IL-15 – JAK1/3 signaling pathway associated with vitiligo recurrence. This positions VC005 to deliver superior long-term efficacy in delaying disease relapse.
Currently, the VC005 tablet is in Phase III clinical trial in China, while the co-developed VC005 topical gel has entered Phase II clinical trial. This IND approval marks the addition of vitiligo as a new indication to VC005’s clinical development pipeline, following its ongoing trials for moderate-to-severe atopic dermatitis and ankylosing spondylitis. The company plans to initiate a Phase II clinical study for this new indication shortly.
About VC005
VC005 is a novel, potent, and highly selective next-generation JAK1 inhibitor independently developed by Jiangsu Vcare. By selectively inhibiting JAK1, the drug reduces inflammatory responses and immune cell activation, making it clinically applicable for treating inflammatory and autoimmune diseases. It is available in two formulations: oral tablets and topical gel.
The oral tablet was developed for multiple autoimmune conditions, including moderate-to-severe atopic dermatitis, ankylosing spondylitis, rheumatoid arthritis, and vitiligo, with the most advanced progress in Phase III trials for moderate-to-severe atopic dermatitis. The topical gel, targeting mild-to-moderate atopic dermatitis, is currently in Phase II trial. Compared to Upadacitinib, a leading marketed drug in the same class, VC005 more selectively reduces JAK2 inhibitory activity (based on in vitro kinase assay results), potentially alleviating safety concerns associated with excessive JAK2 inhibition in clinical settings.
About Vitiligo
Vitiligo is a localized or generalized depigmentary disorder caused by a reduction or disappearance of tyrosinase activity in melanocytes within the skin and hair follicles, resulting in decreased or loss of melanin granule production. It is characterized by complete loss of skin and mucous membrane pigmentation in a localized or generalized manner. It can occur in any part of the body, commonly on the back of the fingers, wrists, forearms, face, neck, etc.
The worldwide lifetime prevalence was estimated at 0.36% in the general population, affecting an estimated 28.5 million people worldwide.
The target mechanism of VC005 is well-defined. In patients with vitiligo, it works in two ways:
By inhibiting the IFN-γ - JAK1/2 - STAT1 signaling pathway, it blocks keratinocytes from releasing chemokines that recruit more CD8+ T cells.
By inhibiting the IL-15 - JAK1/3 - STAT3/5 signaling pathway, it suppresses the activation of Trm cells.
This is how VC005 aims to achieve disease reversal and delay recurrence.
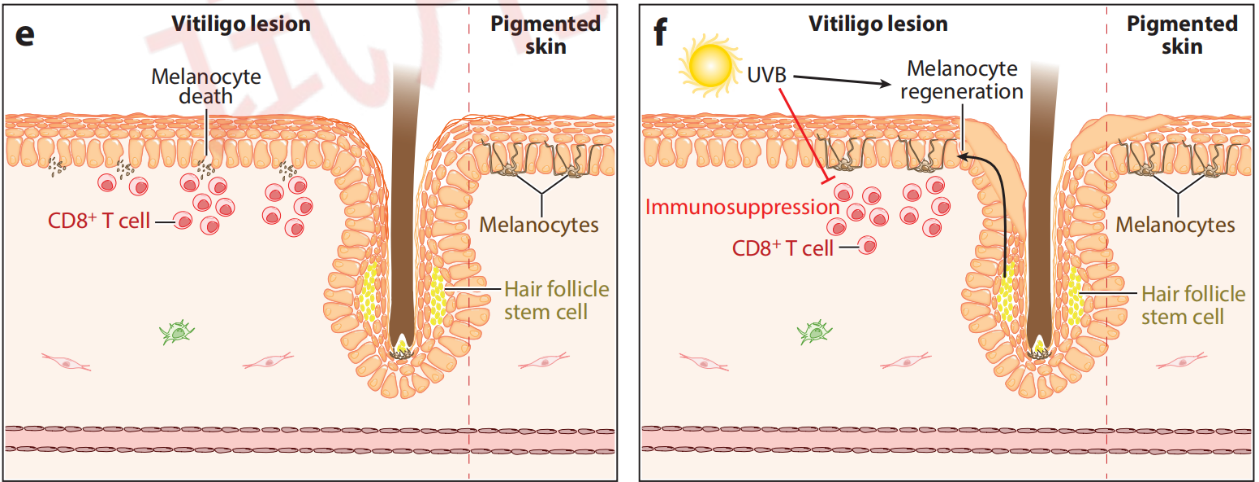
Vitiligo Pathogenesis and Therapeutic Mechanisms
Citing from Frisoli, et al, Vitiligo: Mechanisms of Pathogenesis and Treatment,2020
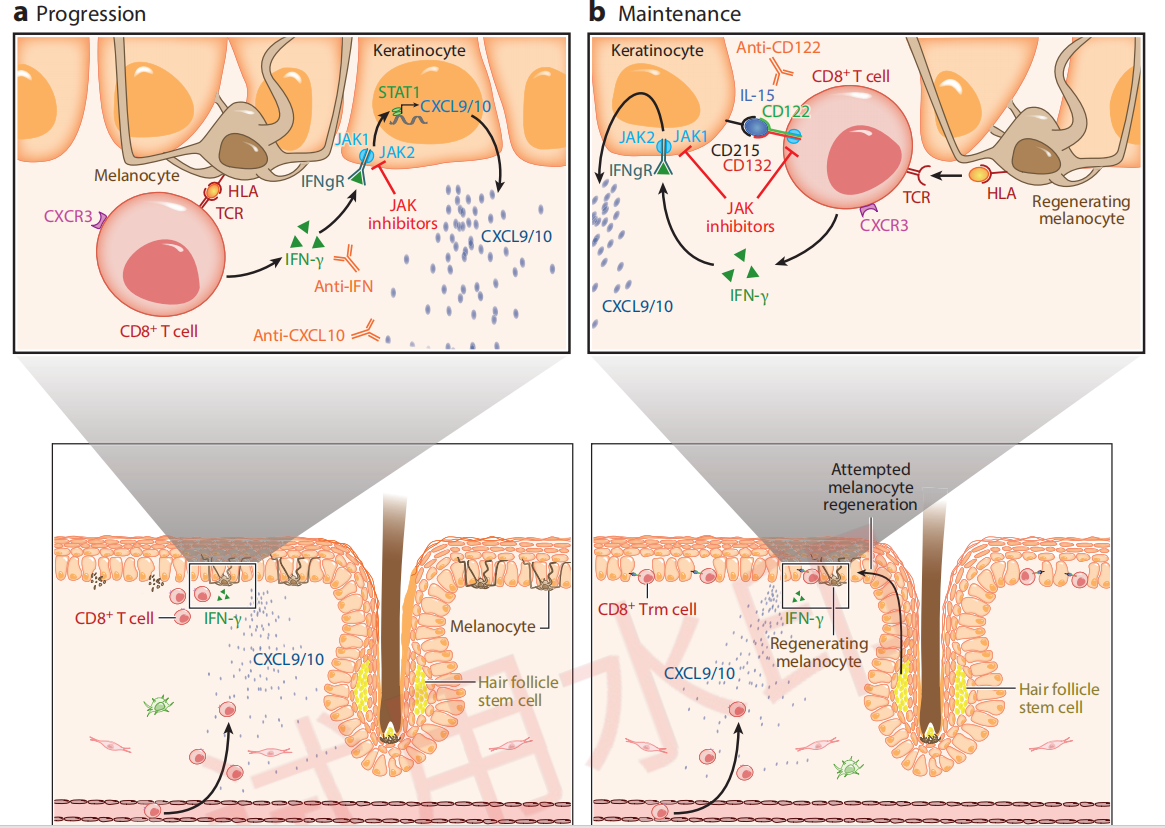
Immunological Mechanisms of Vitiligo Progression and Maintenance
Citing from Frisoli, et al, Vitiligo: Mechanisms of Pathogenesis and Treatment,2020
Previous Page
Related News
05
2024
/
01
Recently, Jiangsu Vcare PharmaTech Co., Ltd. (Jiangsu Vcare) announced the completion of Phase II clinical enrollment for its self-developed second-generation selective JAK1 inhibitor VC005 tablets in the treatment of ankylosing spondylitis (AS). Preliminary blinded analysis from the clinical trial demonstrated favorable efficacy and safety profiles for VC005.
04
2024
/
01
Recently, Jiangsu Vcare Pharmaceutical Technology Co., Ltd. (Jiangsu Vcare) completed the first patient enrollment in the pivotal Phase II clinical trial (Registration Number: CTR20201703) in China for its self-developed next-generation TRK inhibitor, VC004. This clinical trial aims to further evaluate the efficacy and safety of VC004 in treating patients with NTRK gene fusion mutations.
03
2024
/
01
Jiangsu Vcare Submitted an NDA for Anti- platelet Drug Vicagrel to the US
Recently, Jiangsu Vcare Pharmaceutical Technology Co., Ltd. (Jiangsu Vcare) submitted an NDA to the US FDA for Vicagrel capsules for the treatment of acute coronary syndrome (ACS), ischemic stroke, and confirmed peripheral arterial disease. This marks the first NDA submission from Jiangsu Vcare's innovative drug pipeline, representing a significant milestone.
02
2024
/
01
Jiangsu Vcare Secures Over CNY 450 Million in Series C Funding
Recently, Jiangsu Vcare Pharmatech Co., Ltd. (Jiangsu Vcare) closed its Series C financing round, raising over CNY 450 million. The investment consortium included China Life Private Equity Investment, DYEE Capital, Hongyao Technology Capital, existing investor Nanjing Innovation Capital Group, and Hangzhou Huadian Investment.
13
2023
/
12
Jiangsu Vcare Approved to Establish "Provincial-Level Enterprise Technology Center"
Recently, the Jiangsu Provincial Department of Industry and Information Technology released the list of enterprises proposed for recognition as 2023 Provincial-Level Enterprise Technology Centers. Jiangsu Vcare Pharmatech Co., Ltd.(Jiangsu Vcare) has been recognized as a"2023 Provincial-Level Enterprise Technology Center".
10
2023
/
11
Jiangsu Vcare's Next-Generation TRK Inhibitor VC004 Enters Pivotal Phase II Trial
Recently, the initiation of the pivotal Phase II trial for Jiangsu Vcare Pharmaceutical Technology Co., Ltd.'s (Jiangsu Vcare) next-generation TRK inhibitor VC004 project was announced at its first center, the Cancer Hospital Chinese Academy of Medical Sciences. Subsequent recruitment will take place across nearly 30 clinical centers nationwide.

